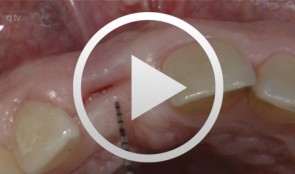
-

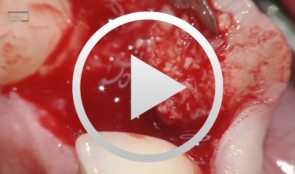
Bone augmentation in the anterior region in preparation for implants
Grunder, UeliOutline: - Incision technique/flap mobilization - Bone removal with trephine cutter 6 mm - Bone bed preparation with trephine cutter 5 mm - Fixation of the autologous bone - Membrane adaptation - Introduction of the replacement material - Attachment of the membrane (with nails) - Introduction of a second membrane - Flap mobilization - Flap closure List of materials: - Trephine cutters (Biomet/3i, Palm Beach Gardens, Florida, USA) - Fixation screws (Biomet Microfi xation, Jacksonville, FL, USA) - e-PTFE membrane (Gore-Tex® reinforced, WL Gore, Flagstaff, AZ, USA ) - Mineralized collagen bone replacement material (Bio-Oss® Collagen, Geistlich Pharma AG, Wolhusen, Switzerland) - Collagen membrane (Biogide, Geistlich Pharma, Wolhusen, Switzerland) -

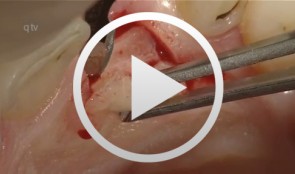
Anterior implant with concurrent bone augmentation (GBR)
Grunder, UeliOutline: - Incision technique/flap mobilization - Implant insertion - Membrane adaptation - Introduction of the replacement material - Attachment of the membrane - Introduction of the second membrane - Flap mobilization - Flap closure List of materials: Titanium implant (Thommen Medical, Waldenburg, Switzerland); e-PTFE membrane (Gore-Tex® reinforced, WL Gore, Flagstaff, AZ, USA); Mineralized collagen bone replacement material (Bio-Oss® Collagen, Geistlich Pharma, Wolhusen, Switzerland); Collagen membrane (Biogide, Geistlich Pharma, Wolhusen, Switzerland) -

Immediate placement of a NobleActive implant in a patient with a pronounced hard-tissue and soft-tissue defect
Nölken, RobertOutline - Modified tubed flap - NobelActive implant placement - Flapless facial bone augmentation - Immediate provisionalization Materials Checklist: NobelActive™ Surgery Kit Twist drill ø 2, 7-15 mm Twist drill ø 2, 10-18 mm Twist step drill ø 2.4/2.8, 7-15 mm Twist step drill ø 2.4/2.8, 10-18 mm Twist step drill ø 3.2/3.6, 7-15 mm Twist step drill ø 3.2/3.6, 10-18 mm Surgical driver NobelActive™ Man torque wrench, surgical NobelActive™ Internal RP implant Procera® esthetic abutment, NobelActive™ Internal Implant replica, NobelActive™ Internal RP Impression coping, open tray, NobelActive™ Internal RP Protect analog, NobelActive™ Internal. -
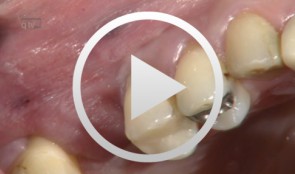

Augmentation at site 16 using the SonicWeld Rx System
Iglhaut, Gerhard M. -
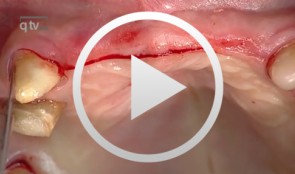

Preimplant Augmentation Procedures to Improve the Hard Tissue Situation in the Upper Anterior Region
Mayer, MatthiasContents - Flap design according to the layering technique - Piezoelectric bone surgery - Bone spreading with osteotomes - Augmentation and suturing Materials Checklist: Piezo surgery unit (ADS); Osteotome (Altatec GmbH); Surgical tray, individual. -
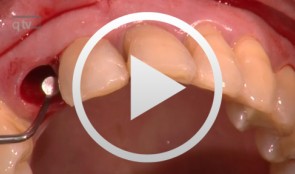

Immediate Function with NoblePerfect Implants
Nölken, RobertProcedure: - Extraction of upper front teeth not suitable for preservation - Immediate loading of four NobelPerfectTM implants - Intraoperative registration of implant position - Flap-free Vestibular Bone Augmentation - Connective Tissue Graft for Periodontal Regeneration - Immediate Loading of temporary implants on the day of surgery - Definitive restoration 6 months later Materials: NobelPerfect Groovy Implants RP and NP; NobelPerfect Temporary Abutments RP and NP; NobelPerfect Implant Replicas RP and WP; Ribbond, Bondable Reinforcement Ribbon; Ethilon 5-0 FS3 Suture Material; Astra Bonetrap Bone Collector. -
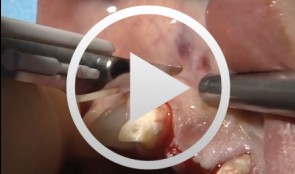

Bone Transplantation with Systemized Armamentarium
Streckbein, Roland -

Regenerative Treatment on Tooth 14 und 24
Eickholz, PeterProcedure: - Incision - Flap Design - Removal of the granulation tissue - Application of the PrefGel on the root surface - Application of the Enamel -Matrix -Protein (Emdogain) - Suture (Offset-Suture) - Identical procedure on the opposite side (1st quadrant) Materials: Retractor Micro Surgical Scalpel Handle Mini Scalpel Blades 4 x Gracey Curettes Periosteal Trombelli Periosteal Prichard Microsurgical Needle Holder Castroviejo Scissors Tweezers Microsurgical Tweezers Gore Tex CV-5 Sutures Gore Tex CV-6 Sutures Emdogain 0,7 ml PrefGel -

-


BIO-ACTIVATION OF DEPROTEINISED BOVINE BONE MINERAL (DBBM) AND NON-CROSS-LINK COLLAGEN MEMBRANES BY THE USE OF GROWTH FACTORS EXTRACTED FROM FRESH AUTOGENOUS BONE CHIPS
Objectives: Autogenous bone grafts are the gold standard for bone augmentation procedures with the ability to release growth factors. These growth factors can be isolated into a "bone-conditioned medium" (BCM). No effort has been made to utilise the growth factors from fresh bone chips in combination with biomaterials to improve bone regeneration. This study aimed to investigate the ability of collagen barrier membranes and DBBM treated with BCM to affect cell behaviour. Methods: Cortical bone chips were harvested from fresh pig mandibles with a bone scraper and placed into plastic dishes containing serum-free culture medium (5g of bone chips per 10mL of medium) for 24 hours. Natural collagen membranes (Bio-GideTM/®) were incubated with BCM for various times. Membranes were also (i) incubated for 4 hours with recombinant TGF-β1; (ii) exposed to ultraviolet light prior to BCM incubation; (iii) pre-wetted for 15 minutes with phosphate buffer saline (PBS) prior to BCM incubation; or (iv) dried and stored at room temperature for 7 days after BCM incubation. After incubation, the membranes were vigorously washed with PBS. DBBM particles (Bio-OssTM/®) were coated with BCM for 5 minutes prior to cell seeding. Gingival fibroblasts or bone-marrow-derived stromal cells (ST2 cells) were seeded on the collagen membranes and DBBM particles, respectively. Messenger RNA levels of BCM target genes were analysed by qRT-PCR using adrenomedullin (ADM), pentraxin 3 (PTX-3), interleukin 11 (IL-11) and proteoglycan-4 (PRG-4). The morphology and viability of cells seeded onto collagen membranes was evaluated. Further, DBBM with and without a BCM coating was compared in terms of cell recruitment, adhesion, proliferation and qRT-PCR for osteoblast differentiation markers (including Runx2, COL1A2, ALP and OCNAlizarin red stain was used to assess mineralisation. The student‘s t-test was used for analysis. Results: Incubation of collagen membranes with BCM for at least 1 minute reduced fibroblast ADM and PTX-3 expression, and increased IL-11 and PRG-4 expression. The four different membrane treatments (i–iv) also provoked significant changes in gene expression. Likewise, conditioned medium from demineralised bone chips caused similar changes in gene expression compared to BCM. BCM did not alter the viability or morphology of gingival fibroblasts on collagen membranes. Coating BCM on DBBM particles improved cell migration of ST2 cells and led toa two-fold increase in cell adhesion. No significant increase in cell proliferation was observed, but BCM significantly increased mRNA levels of COL1a2, ALP and OCN at 3 days post-seeding. A three-fold increase in alizarin red staining was observed on DBBM particles that were pre-coated with BCM. Conclusions: Collagen membranes rapidly adsorb the TGF-β activity of bone chips, and pre-coating DBBM with BCM enhances the osteoconductive properties of DBBM by mediating osteoblast recruitment, attachment and differentiation towards an osteoblast phenotype. These cellular effects of BCM, in combination with biomaterials, might contribute to the overall process of guided bone regeneration. Further animal studies are needed to characterise the added benefit of BCM as an autogenous growth factor for combination therapies.