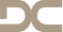-

Immediate placement of a NobleActive implant in a patient with a pronounced hard-tissue and soft-tissue defect
Nölken, RobertOutline - Modified tubed flap - NobelActive implant placement - Flapless facial bone augmentation - Immediate provisionalization Materials Checklist: NobelActive™ Surgery Kit Twist drill ø 2, 7-15 mm Twist drill ø 2, 10-18 mm Twist step drill ø 2.4/2.8, 7-15 mm Twist step drill ø 2.4/2.8, 10-18 mm Twist step drill ø 3.2/3.6, 7-15 mm Twist step drill ø 3.2/3.6, 10-18 mm Surgical driver NobelActive™ Man torque wrench, surgical NobelActive™ Internal RP implant Procera® esthetic abutment, NobelActive™ Internal Implant replica, NobelActive™ Internal RP Impression coping, open tray, NobelActive™ Internal RP Protect analog, NobelActive™ Internal. -

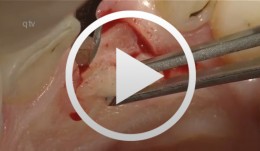
Augmentation at site 16 using the SonicWeld Rx System
Iglhaut, Gerhard M. -

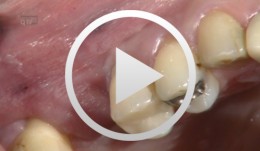
Preimplant Augmentation Procedures to Improve the Hard Tissue Situation in the Upper Anterior Region
Mayer, MatthiasContents - Flap design according to the layering technique - Piezoelectric bone surgery - Bone spreading with osteotomes - Augmentation and suturing Materials Checklist: Piezo surgery unit (ADS); Osteotome (Altatec GmbH); Surgical tray, individual. -
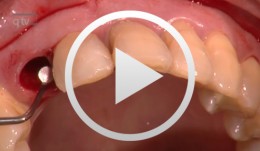

Immediate Function with NoblePerfect Implants
Nölken, RobertProcedure: - Extraction of upper front teeth not suitable for preservation - Immediate loading of four NobelPerfectTM implants - Intraoperative registration of implant position - Flap-free Vestibular Bone Augmentation - Connective Tissue Graft for Periodontal Regeneration - Immediate Loading of temporary implants on the day of surgery - Definitive restoration 6 months later Materials: NobelPerfect Groovy Implants RP and NP; NobelPerfect Temporary Abutments RP and NP; NobelPerfect Implant Replicas RP and WP; Ribbond, Bondable Reinforcement Ribbon; Ethilon 5-0 FS3 Suture Material; Astra Bonetrap Bone Collector. -
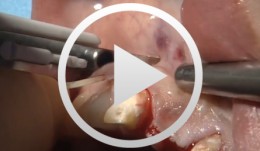

Bone Transplantation with Systemized Armamentarium
Streckbein, Roland -
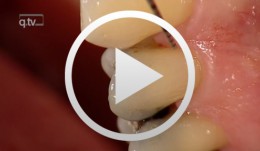

Regenerative Treatment on Tooth 14 und 24
Eickholz, PeterProcedure: - Incision - Flap Design - Removal of the granulation tissue - Application of the PrefGel on the root surface - Application of the Enamel -Matrix -Protein (Emdogain) - Suture (Offset-Suture) - Identical procedure on the opposite side (1st quadrant) Materials: Retractor Micro Surgical Scalpel Handle Mini Scalpel Blades 4 x Gracey Curettes Periosteal Trombelli Periosteal Prichard Microsurgical Needle Holder Castroviejo Scissors Tweezers Microsurgical Tweezers Gore Tex CV-5 Sutures Gore Tex CV-6 Sutures Emdogain 0,7 ml PrefGel -

-


BIO-ACTIVATION OF DEPROTEINISED BOVINE BONE MINERAL (DBBM) AND NON-CROSS-LINK COLLAGEN MEMBRANES BY THE USE OF GROWTH FACTORS EXTRACTED FROM FRESH AUTOGENOUS BONE CHIPS
Objectives: Autogenous bone grafts are the gold standard for bone augmentation procedures with the ability to release growth factors. These growth factors can be isolated into a "bone-conditioned medium" (BCM). No effort has been made to utilise the growth factors from fresh bone chips in combination with biomaterials to improve bone regeneration. This study aimed to investigate the ability of collagen barrier membranes and DBBM treated with BCM to affect cell behaviour. Methods: Cortical bone chips were harvested from fresh pig mandibles with a bone scraper and placed into plastic dishes containing serum-free culture medium (5g of bone chips per 10mL of medium) for 24 hours. Natural collagen membranes (Bio-GideTM/®) were incubated with BCM for various times. Membranes were also (i) incubated for 4 hours with recombinant TGF-β1; (ii) exposed to ultraviolet light prior to BCM incubation; (iii) pre-wetted for 15 minutes with phosphate buffer saline (PBS) prior to BCM incubation; or (iv) dried and stored at room temperature for 7 days after BCM incubation. After incubation, the membranes were vigorously washed with PBS. DBBM particles (Bio-OssTM/®) were coated with BCM for 5 minutes prior to cell seeding. Gingival fibroblasts or bone-marrow-derived stromal cells (ST2 cells) were seeded on the collagen membranes and DBBM particles, respectively. Messenger RNA levels of BCM target genes were analysed by qRT-PCR using adrenomedullin (ADM), pentraxin 3 (PTX-3), interleukin 11 (IL-11) and proteoglycan-4 (PRG-4). The morphology and viability of cells seeded onto collagen membranes was evaluated. Further, DBBM with and without a BCM coating was compared in terms of cell recruitment, adhesion, proliferation and qRT-PCR for osteoblast differentiation markers (including Runx2, COL1A2, ALP and OCNAlizarin red stain was used to assess mineralisation. The student‘s t-test was used for analysis. Results: Incubation of collagen membranes with BCM for at least 1 minute reduced fibroblast ADM and PTX-3 expression, and increased IL-11 and PRG-4 expression. The four different membrane treatments (i–iv) also provoked significant changes in gene expression. Likewise, conditioned medium from demineralised bone chips caused similar changes in gene expression compared to BCM. BCM did not alter the viability or morphology of gingival fibroblasts on collagen membranes. Coating BCM on DBBM particles improved cell migration of ST2 cells and led toa two-fold increase in cell adhesion. No significant increase in cell proliferation was observed, but BCM significantly increased mRNA levels of COL1a2, ALP and OCN at 3 days post-seeding. A three-fold increase in alizarin red staining was observed on DBBM particles that were pre-coated with BCM. Conclusions: Collagen membranes rapidly adsorb the TGF-β activity of bone chips, and pre-coating DBBM with BCM enhances the osteoconductive properties of DBBM by mediating osteoblast recruitment, attachment and differentiation towards an osteoblast phenotype. These cellular effects of BCM, in combination with biomaterials, might contribute to the overall process of guided bone regeneration. Further animal studies are needed to characterise the added benefit of BCM as an autogenous growth factor for combination therapies. -


A COMPARATIVE ANALYSIS OF DEMINERALISED FREEZE-DRIED BONE (DFDBA), FRESH FROZEN BONE ALLOGRAFT (FFBA) AND AUTOGENOUS BONE GRAFT (AU)—A HISTOLOGIC STUDY IN RABBITS
Objectives: There are different clinical applications for bone grafts in alveolar reconstructions and difficulties in achieving vertical osseous increase. This study was to make a comparative histological evaluation of DFDBA, FFBA, AU and blood clot (CO) on vertical guided bone regeneration (GBR) in rabbit calvaria. Methods: Nine rabbits were used. One was the primary bone graft donor and eight were GBR models, whereby 32 titanium cylinders were fixed to the calvaria and randomly filled with DFDBA, FFBA, AU or CO. The animals were killed 13 weeks later and the contents of the cylinders were analysed histomorphologically and histomorphometrically to quantify the total area (AT) of newly formed tissue, new bone (NB) and residual graft (RG) particles. Results: Mean values of AT were significantly higher for DFDBA and FFBA in the order DFDBA = FFBA > AU > CO. New bone formation with DFDBA and FFBA was better than with AU or CO. There were more RG particles in the DFDBA models, in the order FFBA > DFDBA = AU = CO (p values Conclusions: Allografts containing DFDBA and FFBA can be considered beneficial for achieving new vertical bone formation. -


STEM CELL BONE ALLOGRAFTS IN MAXILLARY SINUS AND RIDGE AUGMENTATION – REPORT OF A CASE
Objectives: To evaluate the use of an allograft cellular matrix containing live stem cells for maxillary sinus and ridge augmentations. Methods: Maxillary sinus and ridge augmentations were performed using an allograft cellular matrix containing live stem cells. The post-operative results were evaluated by CT scans and peri-apical radiographs. Sinus augmentation was evaluated after 10 weeks. Radiographic bone tomography was similar to that of the native bone and the ridge augmentation resulted in a vertical ridge augmentation of 3–4mm. The cellular matrix was supplied by Brockton, MA and processed by AlloSource, Centennial, CO. Results: Following healing and approximately 10 weeks following surgery, an additional CT scan was taken. This showed that the native and augmented bone was of an adequate width for supporting an implant. Radiography revealed that the augmented bone had a similar texture to native bone, indicating formation of mature bone. The scan also revealed downward growth of the bone in a vertical direction, overlapping the crest of the native pre-maxillary bone. This was not attempted during the surgical procedure, and was a particular cause for concern. Conclusions: This use of allograft mesenchymal stem cells has been shown to be a reliable method for ridge augmentation, especially in the vertical direction in areas of severe ridge atrophy. Further studies are needed to support this finding in a more guided manner, especially for vertical ridge augmentation. -

-

Cell-to-Cell Communication: Periodontal Regeneration
Stadlinger, Bernd / Terheyden, HendrikUnlike bone, which undergoes remodeling by resorption followed by bone apposition, teeth are not subject to physiological remodeling. They are remarkably resistant to physiological remodeling processes. The reason for this is found in the periodontium and cementum. The mechanisms that take effect in this area and the effectiveness of the periodontal system - consisting of gingiva, alveolar bone, periodontium and cementum - after injuries will be visualized in the new scientific 3D film, "Periodontal Regeneration".The visualization of these complex processes is of great interest for academic teaching as well as for the clinician and general practitioner as the cellular interactions are presented in the context of four phases. For the first time scanning electron microscopic images of real cells will be visualized allowing the comparison of their characteristics with computer animated simulations. DVD 1: Expert Version approx. 14 minutes DVD 2: Public Version approx. 13 minutes Outline: - Cementum formation - Orthodontic tooth movement - Trauma and periodontitis - Periodontal regeneration